he most ‘perfect’ scenario for dementia screening would be to identify dementia in a group of individuals who have absolutely no symptoms might have subtle changes on their volumetric MRI scans, or might have weird protein fragments in their cerebrospinal fluid through an invasive lumbar culture; and then come up with a reliable way to stop it in its tracks The cost, practicality and science behind this prohibit this approach.
There are well defined criteria for screening, such as the “Wilson Jungner criteria“. Prof Carol Brayne from the University of Cambridge has warned against the perils of backdoor screening of dementia, and the need for evidence-based policy, publicly in an article in the British Medical Journal:
“As a group of clinical and applied researchers we urge governments, charities, the academic community and others to be more coordinated in order to put the policy cart after the research horse. Dementia screening should neither be recommended nor routinely implemented unless and until there is robust evidence to support it. The UK can play a unique role in providing the evidence base to inform the ageing world in this area, whilst making a positive difference to the lives of individuals and their families in the future.”
However, a problem has arisen in how aggressively to find new cases of dementia in primary care, and a lack of acknowledgement by some that incentivising dementia diagnosis might possibly have an untoward effect of misdiagnosing (and indeed mislabelling) some individuals, who do not have dementia, with dementia. Unfortunately there are market forces at work here, but the primary consideration must be the professional judgment of clinicians.
Diagnosing dementia
There is no single test for dementia.
A diagnosis of dementia can only be confirmed post mortem, but there are ‘tests’ in vivo which can be strongly indicative of a specific dementia diagnosis (such as brain biopsy for Variant Creutzfeld-Jacob disease or cerebral vasculitis), or specific genetic mutations on a blood test (such as for relatively rare forms of the dementia of the Alzheimer type).
Memory vs non-memory functions in CANTAB
CANTABmobile is a new touchscreen test for identifying memory impairment, being described as a ‘rapid memory test’. The hope is that memory deficits might be spotted quickly in persons attending the National Health Service, and this is indeed a worthy cause potentially. In the rush to try to diagnose dementia quickly (and I have explained above the problem with the term “diagnose dementia”), it is easy to conflate dementia and memory problems. However, I demonstrated myself in a paper in Brain in 1999 using one of the CANTAB tests that patients with behavioural variant frontotemporal dementia (bvFTD) were selectively impaired on tests sensitive to prefrontal lobe function involving cognitive flexibility and decision-making. I demonstrated further in a paper in the European Journal of Neuroscience in 2003 that such bvFTD patients were unimpaired on the CANTAB paired associates learning test.
bvFTD is significant as it is a prevalent form of dementia in individuals below the age of 60. The description given by Prof John Hodges in the current Oxford Textbook of Medicine chapter on dementia is here. Indeed, this chapter cites my Brain paper:
“Patients present with insidiously progressive changes in personality and behaviour that refl ect the early locus of pathology in orbital and medial parts of the frontal lobes. There is often impaired judgement, an indifference to domestic and professional responsibilities, and a lack of initiation and apathy. Social skills deteriorate and there can be socially inappropriate behaviour, fatuousness, jocularity, abnormal sexual behaviour with disinhibition, or theft. Many patients are restless with an obsessive–compulsive and ritualized pattern of behaviour, such as pacing or hoarding. Emotional labiality and mood swings are seen, but other psychiatric phenomena such as delusions and hallucinations are rare. Patients become rigid and stereotyped in their daily routines and food choices. A change in food preference towards sweet foods is very characteristic. Of importance is the fact that simple bedside cognitive screening tests such as the Mini-Mental State Examination (MMSE) are insensitive at detecting frontal abnormalities. More detailed neuropsychological tests of frontal function (such as the Wisconsin Card Sorting Test or the Stroop Test) usually show abnormalities. Speech output can be reduced with a tendency to echolalia (repeating the examiner’s last phrase). Memory is relatively spared in the earl stages, although it does deteriorate as the disease advances. Visuospatial function remains remarkably unaffected. Primary motor and sensory functions remain normal. Primitive refl exes such as snout, pout, and grasp develop during the disease process. Muscle fasciculations or wasting, particularly affecting the bulbar musculature, can develop in the FTD subtype associated with MND.”
Memory tests, mild cognitive impairment and dementia of Alzheimer type
Nobody can deny the undeniable benefits of a prompt diagnosis, when correct, of dementia, but the notion that not all memory deficits mean dementia is a formidable one. Besides, this tweeted by Prof Clare Gerada, Chair of the Royal College of General Practitioners, to me this morning I feel is definitely true,
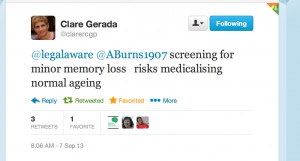
A political drive, almost in total parallel led by the current UK and US governments, to screen older people for minor memory changes could potentially be leading to unnecessary investigation and potentially harmful treatment for what is arguably an inevitable consequence of ageing. There are no drugs that prevent the progression of dementia according to human studies, or are effective in patients with mild cognitive impairment, raising concerns that once patients are labelled with mild cognitive deficits as a “pre-disease” for dementia, they may try untested therapies and run the risk of adverse effects.
The idea itself of the MCI as a “pre-disease” in the dementia of Alzheimer type is itself erroneous, if one actually bothers to look at the published neuroscientific evidence. A mild cognitive impairment (“MCI”) is a clinical diagnosis in which deficits in cognitive function are evident but not of sufficient severity to warrant a diagnosis of dementia (Nelson and O’Connor, 2008).It is claimed that on the CANTABmobile website that:

However, the evidence of progression of MCI (mild cognitive impairment) to DAT is currently weak. It might be attractive to think that MCI is a preclinical form of dementia of Alzheimer Type, but unfortunately the evidence is not there to back this claim up at present: only approximately 5-10% and most people with MCI will not progress to dementia even after ten years of follow-up (Mitchell and Shiri-Feshki, 2009).
An equally important question is also the specificity and sensitivity of the CANTABmobile PAL test. Quite a long explanation is given on their webpage again:
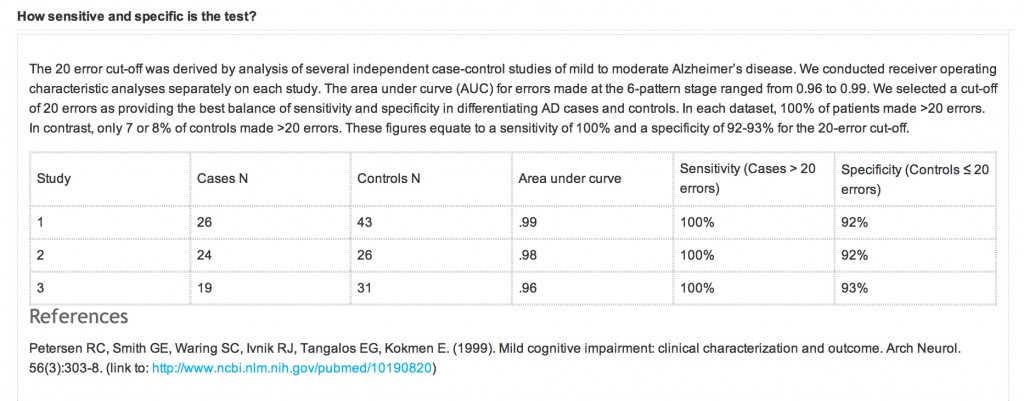
However, the reference that is given is unrelated to the data presented above. What should have appeared there was a peer-reviewed paper analysing sensitivity and sensitivity of the test, across a number of relevant patient groups, such as ageing ‘normal’ volunteers, patients with geriatric depression, MCI, DAT, and so on. A reference instead is given to a paper in JAMA which does not even mention CANTAB or CANTABmobile.
NICE, QOF and indicator NM72
A description of QOF is on the NICE website:
“Introduced in 2004 as part of the General Medical Services Contract, the QOF is a voluntary incentive scheme for GP practices in the UK, rewarding them for how well they care for patients.
The QOF contains groups of indicators, against which practices score points according to their level of achievement. NICE’s role focuses on the clinical and public health domains in the QOF, which include a number of areas such as coronary heart disease and hypertension.
The QOF gives an indication of the overall achievement of a practice through a points system. Practices aim to deliver high quality care across a range of areas, for which they score points. Put simply, the higher the score, the higher the financial reward for the practice. The final payment is adjusted to take account of the practice list size and prevalence. The results are published annually.”
According to guidance on the NM72 indicator from NICE dated August 2013, this indicator (“NM72″) comprises the percentage of patients with dementia (diagnosed on or after 1 April 2014) with a record of FBC, calcium, glucose, renal and liver function, thyroid function tests, serum vitamin B12 and folate levels recorded up to 12 months before entering on to the register The timeframe for this indicator has been amended to be consistent with a new dementia indicator NM65 (attendance at a memory assessment service).
Strictly speaking then QOF is not about screening as it is for patients with a known diagnosis of dementia. If this battery of tests were done on people with a subclinical amnestic syndrome as a precursor to a full-blown dementia syndrome with an amnestic component, it might conceivably be ‘screening’ depending on how robust the actual diagnosis of the dementia of those individuals participating actually is. As with all these policy moves, it is very easy to have unintended consequences and mission creep.
According to this document,
“There is no universal consensus on the appropriate diagnostic tests to be undertaken in people with suspected dementia. However, a review of 14 guidelines and consensus statements found considerable similarity in recommendations (Beck et al. 2000). The main reason for undertaking investigations in a person with suspected dementia is to exclude a potentially reversible or modifying cause for the dementia and to help exclude other diagnoses (such as delirium). Reversible or modifying causes include metabolic and endocrine abnormalities (for example, vitamin B12 and folate deficiency, hypothyroidism, diabetes and disorders of calcium metabolism).
The NICE clnical guideline on dementia (NICE clinical guideline 42) states that a basic dementia screen should be performed at the time of presentation, usually within primary care. It should include:
- routine haematology
- biochemistry tests (including electrolytes, calcium, glucose, and renal and liver function)
- thyroid function tests
- serum vitamin B12 and folate levels.”
It is vehemently denied that primary care is ‘screening’ for dementia, but here is a QOF indicator which explicitly tries to identify reversible causes of dementia in those with possible dementia.
There are clearly issues of valid consent for the individual presenting in primary care.
Prof Clare Gerada has previously warned to the effect that it is crucial that QOF does not “overplay its hand”, for example:
“QOF is risking driving out caring and compassion from our consultations. We need to control it before it gets more out of control – need concerted effort by GPC and RCGP.”
Conclusion
Never has it been more important than to heed Prof Brayne’s words:
“As a group of clinical and applied researchers we urge governments, charities, the academic community and others to be more coordinated in order to put the policy cart after the research horse.”
In recent years, many glib statements, often made by non-experts in dementia, have been made regarding the cognitive neuroscience of dementia, and these are distorting the public health debate on dementia to its detriment. An issue has been, sadly, a consideration of what people (other than individual patients themselves) have had to gain from the clinical diagnosis of dementia. At the moment, some politicians are considering how they can ‘carve up’ primary care, and some people even want it to act as a referral source for private screening businesses. The “NHS MOT” would be feasible way of the State drumming up business for private enterprises, even if the evidence for mass screening is not robust. The direction of travel indicates that politicians wish to have more ‘private market entrants’ in primary care, so how GPs handle their QOF databases could have implications for the use of ‘Big Data’ tomorrow.
With headlines such as this from as recently as 18 August 2013,

this is definitely ‘one to watch’.
Further references
Beck C, Cody M, Souder E et al. (2000) Dementia diagnostic guidelines: methodologies, results, and implementation costs. Journal of the American Geriatrics Society 48: 1195–203
Mitchell, A.J., and Shiri-Feshki, M. (2009) Rate of progression of mild cognitive impairment to dementia -meta-analysis of 41 robust inception cohort studies. Acta Psychiatr Scand, 119(4), pp. 252-65.
Nelson, A.P., and O’Connor, M.G. (2008) Mild cognitive impairment: a neuropsychological perspective, CNS Spectr, 13(1), pp. 56-64.
National Institute for Health and Clinical Excellence (2006) Dementia. Supporting people with dementia and their carers in health and social care. NICE clinical guideline 42
Many thanks to @val_hudson for a useful critical comment about an earlier version of this blogpost.






