In everyday English, a “last chance saloon” means a situation beyond all rational hope.
After decades of working on drugs for Alzheimer’s disease (AD), the best the industry can manage is some drugs which have some effect on symptoms for a few months, but for which there’s no evidence they delay the progression in the long term.
Big Pharma have maintained this deception uptil the very last minute, indeed uptil the patents ran out.
They know they’re now drinking at “the last chance saloon”.
David Cameron backed Maria Miller, and that failed.
He backed the Big Society, and that failed.
Now he has backed research into dementia-busting medications.
After a 16-month inquiry, a verdict was reached on Maria Miller.
Commissioner Hudson found that Miller should have designated the Wimbledon property as her main residence, that she should have reduced her claims by two-sevenths to take account of her parents’ presence and that she overclaimed for interest on the mortgage by around £45,000.
Nonetheless, there was no indication Miller had done anything unlawful or illegal in her deception.
On the other hand, the Japanese drugs company Takeda was fined a record £3.6bn ($6bn) by a federal court in the United States on 8 April 2014 following claims that it concealed a possible link between the drug pioglitazone and bladder cancer.
The fine is the largest to be imposed on any pharmaceutical company.
Takeda’s US partner Eli Lilly, who marketed and sold pioglitazone in the United States between 1999 and 2006, also received a £1.79bn ($3bn) fine.
The diabetes drug pioglitazone, marketed as Actos in the US, received marketing authorisation in Europe in 2000. Actos is marketed and sold in the United Kingdom by Takeda UK Ltd.
Today we found out that Tamiflu doesn’t work so well after all. Roche, the drug company behind it, withheld vital information on its clinical trials for half a decade, but the Cochrane Collaboration, a global not-for-profit organisation of 14,000 academics, finally obtained all the information.
Putting the evidence together, it has found that Tamiflu has little or no impact on complications of flu infection, such as pneumonia.
The huge scandal, of course, is that scandal Roche broke no law by withholding vital information on how well its drug works.
Elsewhere, standardised tobacco packaging is intended to reduce the appeal of tobacco products by removing advertising and increasing the prominence of health warnings.
This measure has strong support from health professionals, particularly as rates of child uptake of smoking are still unacceptably high.
Tobacco industry misrepresentation of the evidence in order to try to block public health interventions by manipulating policy making and public opinion is now well documented.
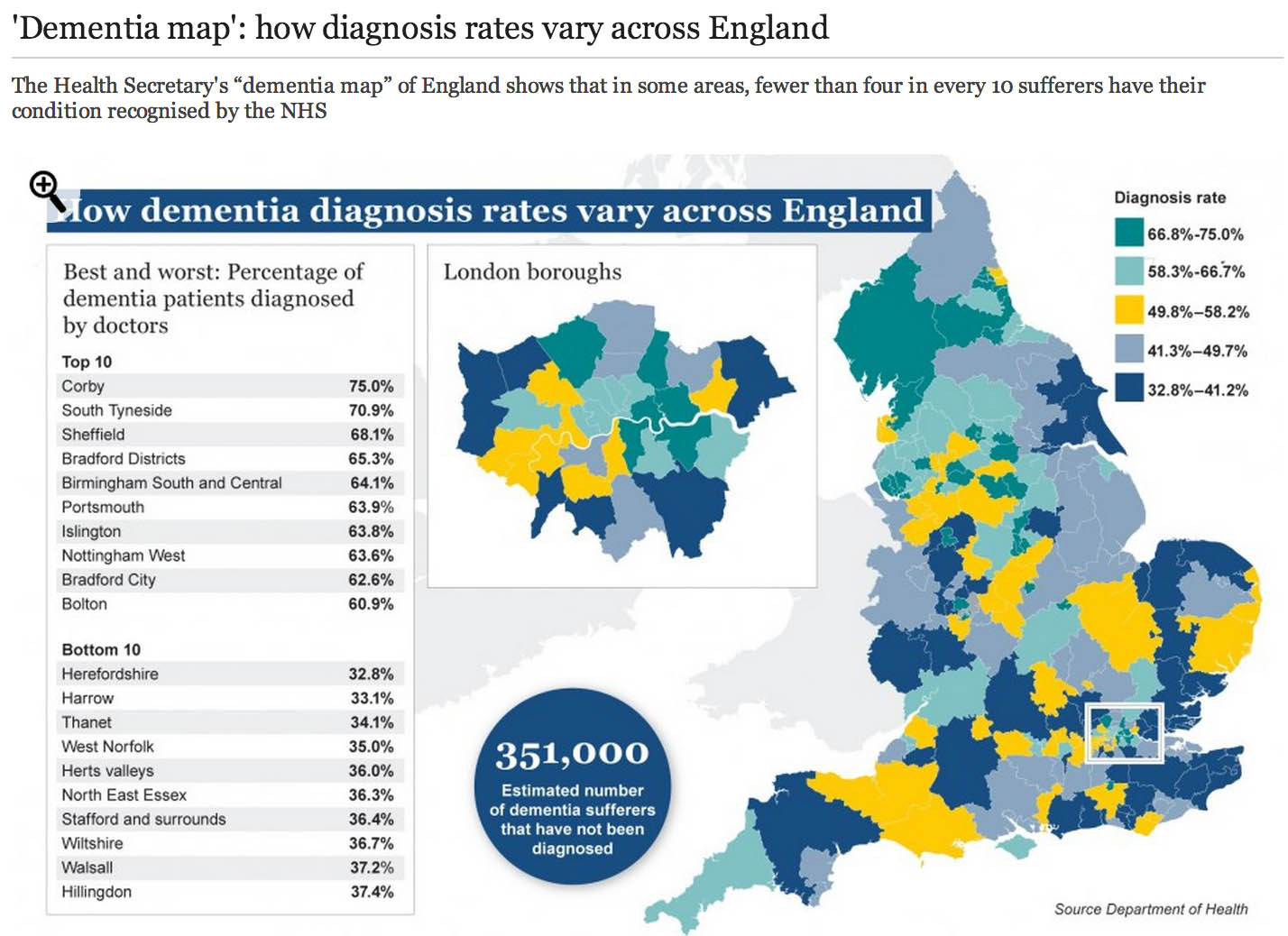
On March 2011, the National Health Service’s National Institute for Health and Care Excellence (NICE) concluded that donepezil hydrochloride (trade name Aricept, Pfizer) could be ‘recommended as (an option) for managing mild as well as moderate AD’.
The conclusion was drawn despite reportedly poor cost efficacy3 and opinions that the use of the drug is a ‘desperate measure’.
The NICE decision was based on two meta-analyses (the second was an update of the first) of randomised controlled trials (RCTs) that demonstrated donepezil’s effect on measures of cognition, behaviour, function and global skills.
Of the 19 studies included, 12 were produced by the companies that manufacture and market donepezil. And a recent study has found that the effect size of donepezil on cognition is larger in industry-funded than independent trials and this is not explained by the longer duration of industry-funded trials.
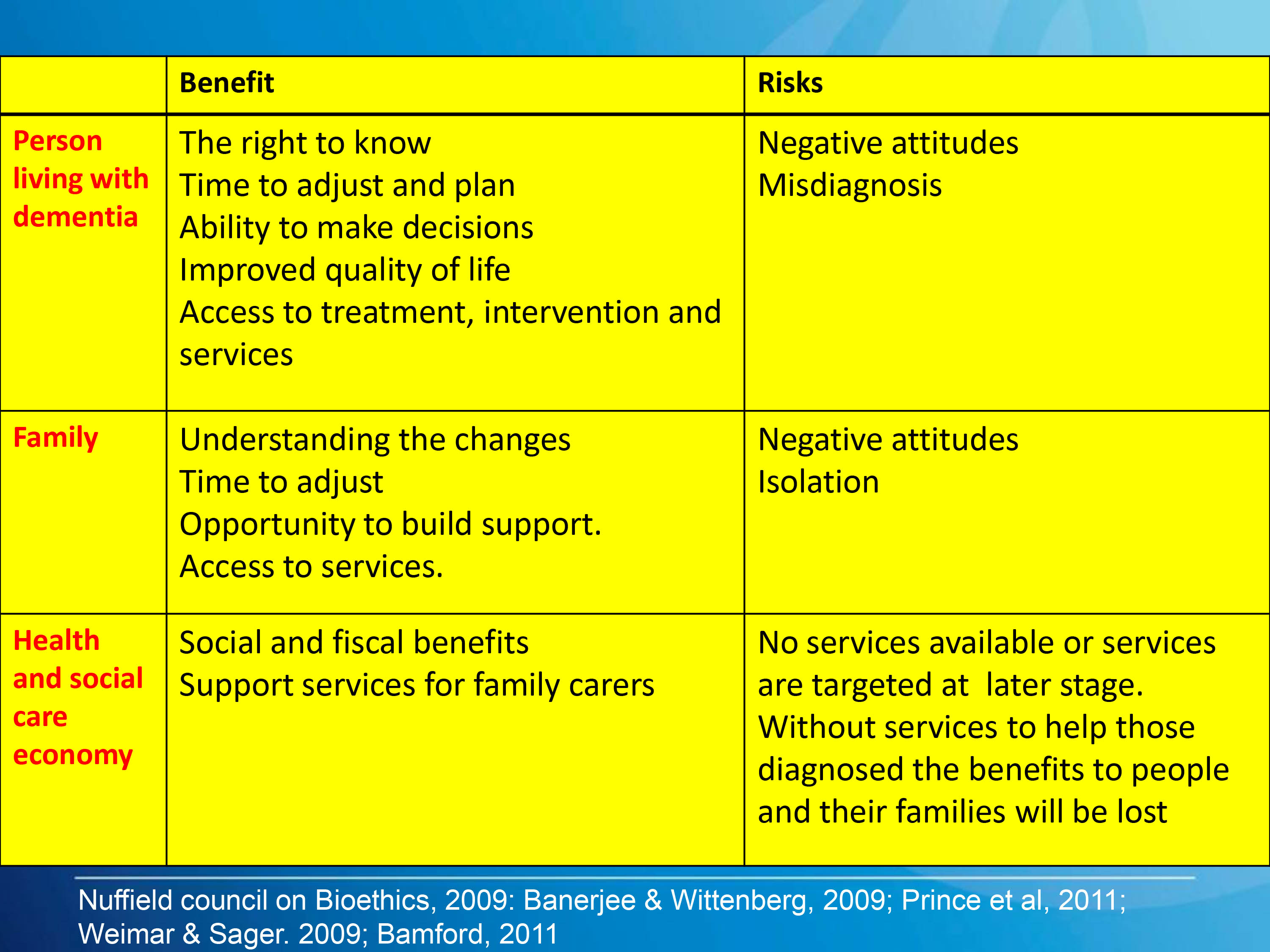
The history of anti-dementia drugs is inglorious. This is significant because every pound spent in flogging this dead horse is a pound denied from current persons living well with dementia.
Tacrine is an oral acetylcholinesterase inhibitor previously used for therapy of Alzheimer disease. Tacrine therapy has been linked to several instances of clinically apparent, acute liver injury.
Because of continuing concerns over safety and availability of other acetylcholinesterase inhibitors, tacrine was withdrawn from use in 2013.
And it is widely reported that current candidate drugs for Alzheimer’s disease are running into problems because of their side effect profile.
Maria Miller may have finally left the ‘last chance saloon’.
But it can’t have escaped Big Pharma, despite ‘the G8 dementia summit’, possibly the largest PR stunt for pharma and research funded by pharma in history, that they are currently drinking there.
The deceptions might be so far be legal.
Maria Miller’s claims might have been hyperbolic; at least she didn’t have a highly staged G8 summit afterwards.
But, as with Maria Miller, the court of public opinion may provide otherwise.